The selection of fluorophores for Immunofluorescent staining depends on:
- Instrument set-up and equipment. For instance, availability of light sources, filter sets, and detection systems.
- Degree of desired color separation for multiple labeling. For example, to achieve good color separation from Alexa Fluor488 or FITC, choose a longer wavelength-emitting fluorophore such as Rhodamin Red-X, Alexa Fluor 594 or Alexa Fluor 647 rather than Cy3.
- Level of antigen expression: for the detection of antigens expressed at low levels chose a brighter fluorophore than for highly expressed antigens.
- Required sensitivity. For example, Alexa Fluor 488, Cy3, Alexa Fluor 594, and Alexa Fluor 647 are brighter than FITC, Cy2, TRITC, Rhodamin-Red X, Texas Red, and Cy5.
Each detection method has different requirements for fluorescent probes
Method:
Condition:
Microscopy
- Sample penetration
- Autofluorescence of sample material
- pH range
- Polar (aqueous) or non-polar (plastic) mounting media
- Antigen abundance ⇒ use bright fluorophores for low expressed analytes
Microscopy (Multiple Labeling)
- Spectral overlap of fluorophores
- Available filtersets (Microscope)
- Antigen abundance ⇒ use bright fluorophores for low expressed analytes
Super-Resolution Microscopy (STORM and STED)
- High emission at STED-Laser wavelength yields high saturation
- Brightness
- Photoswitch
- Photostability
Flow Cytometry
- Large and bright fluorescent proteins (R-PE, APC, PerCP) or Brilliant Violet conjugates are only recommended for labeling surface proteins
- Small fluorescent dyes can be used for surface proteins and intracellular staining.
Western Blot and dot blotting
- Far-red and infrared fluorescent dyes for high sensitivity
- Spectral overlap
Available Fluorochromes for Histo-/Cytochemistry and Flow Cytometry
| Fluorochrome | Amax [nm] | Emax [nm] | Color | Antibody – Dilution | |
| DyLight 350 | DyLight 350 | 353 | 432 | Blue-Violet | 1:100 – 1:800 |
| DyLight 405 | DyLight 405 | 400 | 421 | Blue-Violet | 1:100 – 1:800 |
| BV 421 | Brilliant Violet 421 | 407 | 421 | Blue-Violet | 1:50 – 1:200 |
| AMCA | Aminomethylcoumarin-Acetat | 350 | 450 | Blue | 1:50 – 1:200 |
| BV 480 | Brilliant Violet 480 | 436 | 478 | Blue | 1:50 – 1:200 |
| Cy2 | Carbocyanin | 492 | 510 | Green | 1:50 – 1:200 |
| DyLight 488 | DyLight 488 | 493 | 518 | Green | 1:100 – 1:800 |
| IFKine™ Green | IFKine™ Green | 493 | 518 | Green | 1:50 – 1:1000 |
| Alexa 488 | Alexa Fluor 488 | 493 | 519 | Green | 1:100 – 1:800 |
| FITC | Fluorescein /-Isothiocyanat) | 492 | 520 | Green | 1:50 – 1:200 |
| Cy3 | Indocarbocyanin | 550 | 570 | Yellow | 1:100 – 1:800 |
| IFKine™ Orange | IFKine™ Orange | 555 | 570 | Yellow | 1:50 – 1:1000 |
| TRITC | Tetramethylrhodamin | 550 | 576 | Yellow | 1:50 – 1:200 |
| DyLight 550 | DyLight 550 | 562 | 576 | Yellow | 1:100 – 1:800 |
| R-PE | R-Phycoerythrin | 490/545/566 | 580 | Orange-Red | 1:50 – 1:200 |
| RRX | Rhodamin Red X | 570 | 590 | Orange-Red | 1:50 – 1:200 |
| Alexa 594 | Alexa Fluor 594 | 591 | 614 | Red | 1:100 – 1:800 |
| IFKine™ Red | IFKine™ Red | 591 | 615 | Red | 1:50 – 1:1000 |
| DyLight 594 | DyLight 594 | 593 | 618 | Red | 1:100 – 1:800 |
| Texas Red | Texas Red | 596 | 620 | Red | 1:50 – 1:200 |
| DyLight 633 | DyLight 633 | 638 | 658 | Red | 1:100 – 1:800 |
| APC | Allophycocyanin | 650 | 660 | Near-Infrared | 1:50 – 1:200 |
| Alexa 647 | Alexa Fluor 647 | 651 | 667 | Near-Infrared | 1:100 – 1:800 |
| Cy5 | Indodicarbocyanin | 650 | 670 | Near-Infrared | 1:100 – 1:800 |
| PerCP | Peridinin-Chlorophyll | 482 | 676 | Near-Infrared | 1:50 – 1:200 |
| Alexa 680 | Alexa Fluor 680 | 684 | 702 | Near-Infrared | 1:100 – 1:800 |
| DyLight 680 | DyLight 680 | 692 | 712 | Near-Infrared | 1:100 – 1:800 |
| DyLight 800 | DyLight 800 | 777 | 794 | Infrared | 1:100 – 1:800 |
| Alexa 790 | Alexa Fluor 790 | 792 | 803 | Infrared | 1:100 – 1:800 |
The fluorescent dyes Cy2 and DyLight 488, or Cy5 and DyLight 649 which have been available in the past, have been replaced by the Alexa Fluor dyes 488 and 647 respectively. Similarly DyLight 594 has been replaced by Alexa Fluor 594. In the range of orange fluorescence emission, the bright and stable Cy3 conjugates from Jackson ImmunoResearch are available. Further, DyLight conjugates are now avaible from ImmunoReagents.
Sample protocol triple labeling
Focus: fluorescent dyes
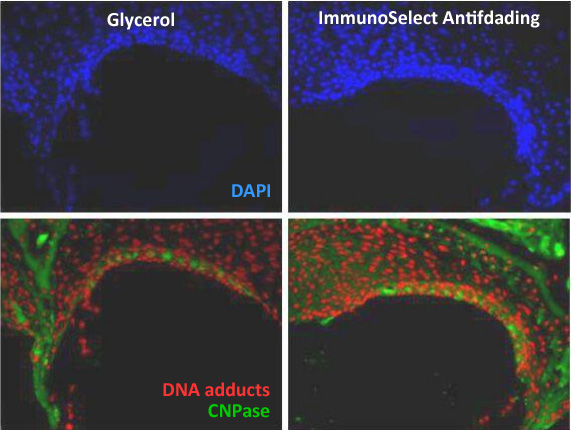
Optimal mounting increases fluorescence stability and signal clarity
As universal mounting medium ImmunoSelect® Antifading Mounting Medium is a perfect tool for multicolor labeling with different fluorescence dyes to suppress photobleaching and preserve signal brightness. The medium is compatible with all dyes and is specifically formulated to eliminate the frequent problem of autofluorescence of mounting media. A strong initial fluorescence combined with very good anti-fading properties and no autofluorescence in the relevant spectral guarantees clear signals with high contrast.
Saving rare samples during staining
Effective immobilization of cells and tissues prevent the loss of valuable sample material during washing and staining steps. Simply dropping sample material onto Immunoselect®Adhesive Slides allows quick and strong fixing without the need of additional centrifugation, drying or fixation. The innovative adhesive coating of the slides reacts with the natural surface structures on cells and tissues and anchors the later permanently on the glass surface via different types of adhesive forces. Even in most extreme conditions Immunoselect® Adhesive Slides prevent samples to be detached and fully preserve their antigenicity.
• Very fast adhesion of cells and tissue sections with high retainment >95%
• New alternative to Polylysine and other adhesion techniques
• No cytospins or smears necessary, simply drop cells and let them float down
• Cell adhesion resistent to heating, staining and denaturation procedures
• No cell loss even at harsh cytological staining procedures
• Superb retainment of cell and tissue morphology.
Fluorescent Conjugates of Secondary Antibodies
All secondary antibodies from Jackson ImmunoResearch Labs are isolated from antisera by immunoaffinity chromatography using antigens coupled to agarose beads. A proprietary elution process (AffiniPure) is used to dissociate antibodies from the antigen in a gentle process which provides antibodies with excellent binding properties.
Further adsorption against serum proteins of different species as well as against IgG, IgM, IgA and IgG-fragments eliminates cross-reactivity and ensures the Ig-specific reactivity of the secondary antibodies.
Apart from the specific binding properties of the antibody (activity, specificity, cross-reactivity), the detection level of any fluorophore-antibody conjugate depends on brightness and photostability of the dye and the optimal dye per antibody ratio. For the production of fluorescent conjugates with optimal binding properties, a molar saturation curve vs. fluorescence intensity, antibody activity, background level, and/or other parameters has been established for each dye to optimize the level of antibody detection and minimize background.
Fluorescence labeled antibodies from Jackson ImmunoResearch Labs are provided freeze-dried in 0.01M Sodium Phosphate/ 0.25M NaCl, pH 7.6 with 15 mg/ml BSA/ 0.05% Sodium Azide. Before first use, reconstitute conjugate with aqua dest. according to the instructions in the data sheet.
Alexa Fluor®, Rhodamin Red™-X and Texas Red® are registered trademarks of Life Technologies, Inc.*. Cy™ is a trademark of GE Healthcare (Patent 5.268.486). Brilliant Violet™ is a trademark of Sirigen Inc., a Becton, Dickinson and Company affiliate. DyLight™ Fluorescent Dyes is a trademark of Thermo Fisher Scientific. The use, manufacture, trading, or sale of these dyes or products containing these dyes is sold pursuant to license agreements of Jackson ImmunoResearch Laboratories, Inc. with the mentioned companies for use of their fluorescent dye technologies.
* The sale of these dyes or products containing these dyes is expressly conditioned on the buyer not using them for any other/further commercial purpose (such as manufacturing, resale, diagnostics, therapy, providing services a. o.). For information on purchasing a license to this product for purposes other than research, contact: Life Technologies Corporation, 5791 Van Allen Way, Carlsbad, CA 92008.