Fluorophores conjugated to secondary antibodies or streptavidin enable a quick and easy detection of analytes. There is a broad range of fluorescent dyes available, each with individual characteristics. Here, you can learn more about Jackson ImmunoResearch secondary antibody conjugates that can be used in various immunoassays (e.g. Flow Cytometrie, Microscopy, Western Blot and ELISA).
This alphabetical list of fluorophores does only contain dyes that are used by us or our partners to manufacture conjugates and is not an exhaustive list of all available fluorophores.
Alexa Fluor® dyes
Alexa Fluor® dyes produce conjugates of intense fluorescence and extreme photostability, making them superior to most other spectrally similar fluorescent dyes. Due to the high absorbance at wavelengths of maximal output of common excitation sources and the low tendency to self-quenching of fluorescence by attachment to proteins, bioconjugates of Alexa Fluor dyes show a bright and extraordinarily stable fluorescence. The fluorescence intensity is insensitive to variations in pH value over a broad range (pH 4 – 10). The good water solubility of the dyes makes their conjugates resistant to precipitation and aggregation preventing problems with background staining. Alexa Fluor dyes are closely related to DyLight dyes because of structural similarities.
Alexa Fluor® 488
Alexa Fluor 488 (Amax 493 nm, Emax 519 nm) has absorption and emission spectra comparable to Fluorescein (FITC), but are much brighter, far more photostable and less sensitive to pH changes between pH 4 and 10. Alexa Fluor 488 conjugates are compatible with common fluorescein equipment, settings, or filters and are ideally suitable for all applications in fluorescence microscopy and flow cytometry with high requirements in sensitivity. Alexa Fluor 488 shows more persistent and brighter fluorescence than Fluorescein in epifluorescence microscopes even without the addition of anti-fading agents to aqueous mounting media. In permanent organic mounting media Alexa Fluor 488 is also more fluorescent and long-term stable than FITC. Alexa Fluor 488 is comparable to DyLight 488.
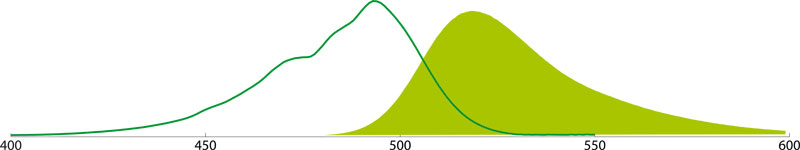
Alexa Fluor® 594
Alexa Fluor 594 conjugates (Amax 591 nm, Emax 614 nm) emit in the red range of the visible light spectrum, are brighter than other red-fluorescing dye conjugates, and allow better color separation from green fluorescent dyes like Alexa Fluor 549, Cy3, or TRITC. They are the best choice for immunofluorescence detection in the far red region of the visible spectrum. Alexa Fluor 594 conjugates are more fluorescent and photostable than Texas Red conjugates, are more water soluble and cause less background. The photostability also depends on the mounting medium: whereas Alexa Fluor 594 conjugates are more stable in ProLong® Gold Antifade (Life Technologies) than Texas Red conjugates, they fade more rapidly in VECTASHIELD® (Vector Laboratories).
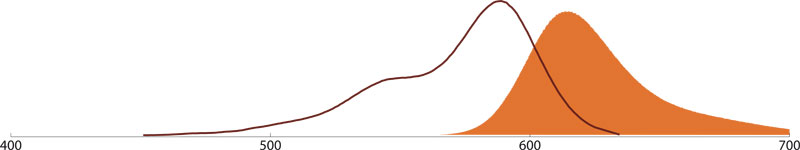
Alexa Fluor® 647
Alexa Fluor 647 conjugates (Amax 651 nm, Emax 667 nm) have almost identical spectral characteristics than Cy5 dye conjugates but are much brighter. Alexa Fluor 647 conjugates are the best choice for confocal laser microscopy and a comparable or even better alternative for APC conjugates for flow cytometry where secondary antibodies emitting in the near-infrared region of the sepctrum are needed.
Due to a wide separation of its emission from that of shorter wavelength-emitting fluorophores Alexa Fluor 647 conjugates can be combined with a variety of other fluorophores and are ideal for multiple labeling in confocal laser scanning microscopy. A significant advantage of using Alexa Fluor 647 is the low autofluorescence of biological samples in this region of the spectrum.
Since the fluorescence emission at 670 nm is not easily visible to the human eye, and can poorly be seen with a conventional epifluorescence microscope, especially as mercury lamps often used in these microscopes do not emit sufficient light within the excitation range for Alexa Fluor 647, conjugates are not recommended for use in epifluorescence microscopy. They are most commonly visualized with a confocal microscope equipped with an appropriate laser for excitation (helium-neon laser (at 633 nm), krypton-ion laser (at 647 nm), diode laser) and a far-red detector.
For flow cytometry Alexa Fluor 647 conjugates are a less expensive and equally bright or even brighter alternative to Allophycocyanin conjugates. Due to the smaller molecular size of Alexa Fluor 647 (1,3 kDa) compared to APC (104 kDa) the resulting antibody conjugates of Alexa Fluor 647 are significantly smaller which make them more suitable for the staining of intracellular antigens in flow cytometry.
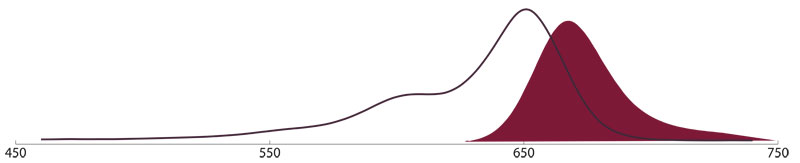
AMCA (Aminomethylcoumarin Acetate)
AMCA conjugates (Amax 350 nm, Emax 450 nm) have been used mostly for multiple labeling since there is minimal fluorescence overlap with green-fluorescing dyes and little or no overlap with longer wavelength-emitting fluorophores. Applications for multiple labeling with this probe include both immunofluorescence microscopy and flow cytometry. Due to its reduced optical performance (fast photobleaching) AMCA is not suggested for single labeling.
Since blue fluorescence is not well detected by the human eye, AMCA-conjugated secondary antibodies should be used with the most abundant antigens in multiple-labeling experiments. For fluorescence microscopy, AMCA can be excited with a mercury lamp and observed using a UV filter set. Ways of improving the visibility of AMCA include dark adapting the eyes, using fluorite instead of glass objectives, avoiding mounting media that absorb UV light (such as plastic-based media), and capturing photographic images with blue-sensitive CCD cameras.
AMCA fades rapidly in conventional epifluorescence and confocal microscopy, and therefore it should be used with mounting media containing an anti-fading agent such as ImmunoSelect®, or n-propyl gallate (2 % n-propyl gallate in 80-90 % glycerol / PBS (pH 7.2 – 9.0)). For flow cytometry, AMCA can be excited with a mercury lamp, or with an argon ion laser which emits some lines in the UV .
AMCA is not suggested for single labeling in one-photon microscopy because of its relatively weak signal and rapid fading. However, AMCA has been found to be a bright and photostable dye for 2-photon microscopy.
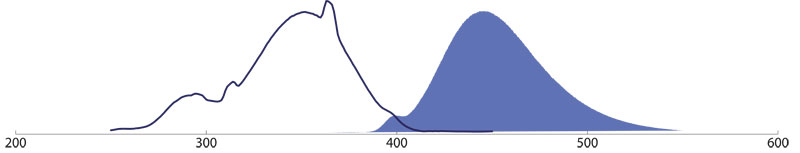
Brilliant Violet™
BD Brilliant Violet conjugates BV421™ and BV480™ provide users with more options in the blue channel. Up to 5 fluorescent dyes can be used in one multiple labeling experiment.
Brilliant Violet 421™ (BV421™)
Brilliant Violet 421™-conjugates (Amax 407 nm, Emax 421nm) are especially useful for multiple labeling assays in combination with BV 480™, Alexa Fluor® 488, Rhodamine Red™-X, and Alexa Fluor® 657.
Please note, due to their physical properties all dyes that can be exited in the blue range have a higher tendency to photobleach than dyes that are exited in higher wavelength.
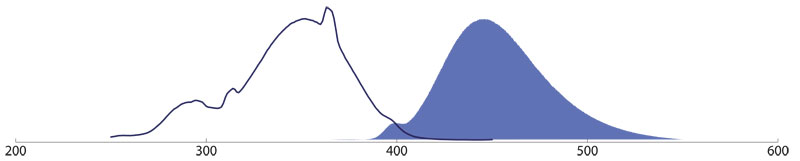
Brilliant Violet 480™ (BV480™)
Brilliant Violet 480™-conjugates (Amax 436 nm, Emax 478 nm) are especially useful for multiple labeling assays in combination with BV 421™, Alexa Fluor® 488, Rhodamine Red™-X, and Alexa Fluor® 657.
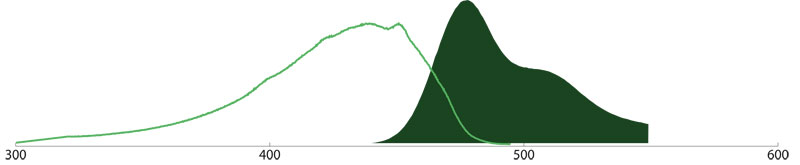
Learn more about BD Brilliant Violet™ secondary antibody conjuagtes.
Cyanine (Cy™) dyes
Cy2™ (Amax 492 nm, Emax 510 nm), Cy3™ (Amax 550 nm, Emax 570 nm) and Cy5™ (Amax 650 nm, Emax 670 nm) belong to a second-generation family of fluorochromes. They are more photostable than for example FITC or TRITC. However, we now recommend conjugates of Alexa Fluor because they have improved properties superior to Cy2 and Cy5 conjugates. Cy2 and Cy5 have been discontinued and replaced by conjugates of Alexa Fluor 488 and Alexa Fluor 647.
Cy2™ (Carbocyanin)
Cy2™ (Carbocyanin) has similar spectral properties than FITC and can be used with the same filtersets. Please note that Cy2 is sensitive against the anti-fading agent N-Phenylendiamine, that can be found in some aqueous mounting media.
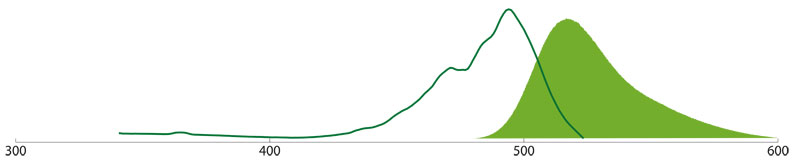
Cy3™ (Indocarbocyanine)
Cy3™ (Indocarbocyanine) is brighter, more photostable, and gives less background than TRITC and most other fluorophores. Cy3 conjugates can be visualized without additional technical equipment with traditional tetramethyl rhodamine (TRITC) filter sets since the excitation and emission spectra are nearly identical to those of TRITC; Cy3-specific filters are also available from various suppliers (e.g. Zeiss filter set 20). Cy3 can be excited to about 50% of maximum with an argon laser (514 nm or 528 nm lines), or to about 75% of maximum with a helium/neon laser (543 nm line) or mercury lamp (546 nm line).
Cy3 is commonly combined with green-fluorescent dyes for double labeling. However, due to the spectral overlap the use of a narrow band-pass emission filter for fluorescein is recommended to minimize Cy3 fluorescence in the FITC filter set. Cy3 can also be paired with near-infrared-fluorescent dyes like Alexa Fluor 647 for multiple labeling when using a confocal microscope equipped with a krypton/argon laser and a far-red detector.
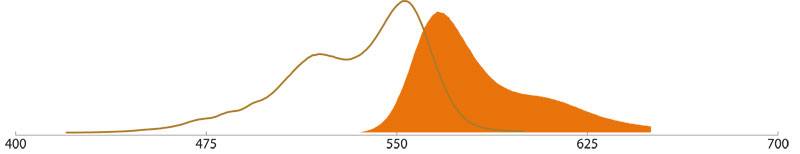
Cy5™ (Indodicarbocyanin)
Due to the minimal overlap with other fluorochromes Cy5™ (Indodicarbocyanin) is mostly used for double or triple stainings.
The emitted fluorescence is not easily detected with the naked eye
especially since high-pressure mercury lamps that are most commonly used
do not emit enough light in the excitation range of Cy5. Cy5 can be
excited with a Krypton/ Argon-Laser (with 98% of the maximum absorption
at 647 nm) or a Helium/Neon-Laser (with 63% of the maximum absorption at
633 nm) or with red diode laser and is mostly used in laser confocal
microscopy.
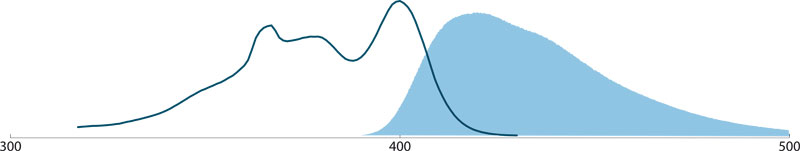
DyLight™ fluorescent dyes
DyLight™ dyes belong to a family of dyes with improved brightness and photostability similar to the Alexa Fluor family of fluorescent dyes. They are highly water soluble and retain their luminosity over a pH range from 4 to 9.
DyLight™ dyes (DyLight 405)
DyLight 405 conjugated Antibodies (Amax 400nm, Emax 421nm) are bright and photostable, but their use is limited to confocal microscopes equipped with a 405 nm laser and appropriate emission filter. Under these conditions, it is possible to perform effective 4-color imaging with good color separation, good photostability, and high sensitivity in both aqueous and permanent mounting media. The combination of DyLight 405, Alexa Fluor 488, Rhodamine Red-X, and Alexa Fluor 647 provides for maximum color separation.
Another 4-color dye combination, which may be equally effective but has less color separation includes Cy3 instead of Rhodamine Red-X. When using aqueous mounting media the addition of n-propyl gallate is recommended to reduce bleaching of fluorescence (mounting medium such as n-propyl gallate-glycerol).
DyLight 405 is not recommended for use in epifluorescence microscopes, nor is it recommended for flow cytometry, because emission filters/excitation sources generally used in these devices are not optimal for DyLight 405. As all fluorophores excited in the blue spectrum DyLight 405 has a higher tendency to photobleach compared to dyes that are excited at higher wavelength.
Here you can find more DyLight secondary antibody conjugates
FITC (Fluorescein Isothiocyanate)
FITC (Amax 492 nm, Emax 520 nm) is traditionally still widely used. The most serious disadvantages are the rapid photobleaching (fading) and the low brightness compared to modern fluorophores. These negative properties can be partially mitigated by the use of an anti-fading agent in the mounting medium.
DTAF (Dichlorotriazinylamino-Fluorescein) and FITC (Fluorescein isothiocyanate) are derivatives of the same fluorescein molecule, therefore sharing identical excitation and emission peaks. Jackson ImmunoResearch uses DTAF (instead of FITC) for conjugation with streptavidin, since fluorescence from FITC is greatly quenched after conjugation with streptavidin. This phenomenon is unique to streptavidin, and is not observed with antibodies. Alternatives with less photobleaching but excitation at the same wavelength are Cy2™ (Carbocyanin) or Alexa Fluor® 488.
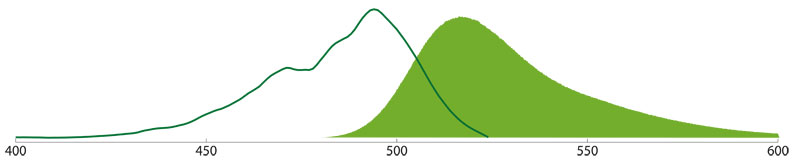
TRITC, Rhodamine Red™-X and Texas Red®
TRITC, Rhodamine Red-X and Texas Red are rhodamine derivatives that fluoresce in the orange and red range of the visible spectrum. Conjugates of these derivatives have different excitation (550, 570, and 596 nm) and emission maxima (570, 590, and 620 nm). All three fluorochromes are suitable for double labeling in combination with FITC or Alexa Fluor 488.
Although TRITC has been used traditionally with FITC for double labeling, better color separation is achieved by using Rhodamin Red-X or Texas Red because of the smaller overlap with the FITC-spectrum. However, it has been reported that use of Texas Red may lead to higher background staining. We now recommend the use of Alexa Fluor 594, instead of Texas Red, because it is clearer, brighter, more photostable, and more hydrophilic than Texas Red. In the orange range, Cy3 provides a brighter alternative to TRITC.
For double labeling with FITC in flow cytometry phycoerythrin (instead of rhodamine) is recommended because both fluorophores can be excited with an argon laser (at 488 nm).
Rhodamine Red™-X
Rhodamine Red-X (Rhod. Red-X, RRX) is particularly useful for three- or even four-color labeling with DyLight 405, Alexa Fluor 488, and Alexa Fluor 647 by using a confocal microscope equipped with a 405 nm-laser and a krypton/argon laser.
Emission from RRX lies about midway between that of Alexa Fluor 488 and Alexa Fluor 647, and shows little overlap with either dye. Moreover, all three fluorochromes can be excited without relevant loss of sensitivity by the krypton-argon laser commonly used in confocal laser microscopes, since the krypton-argon laser emits lines at 488 nm, 568 nm, and 647 nm, all of which are optimal matched for exciting Alexa Fluor 488 (FITC), Rhodamine Red-X and Alexa Fluor 647, respectively. By adding a 405 nm laser four-color labeling is possible using DyLight 405-conjugated secondary antibodies.
In epifluorescence microscopy conjugates of Rhodamine Red-X are particularly suitable used in combination with green-fluorescent dyes (Alexa Fluor 488). Another possibility is the simultaneous use of Rhodamine Red-X conjugated antibodies with blue-fluorescent AMCA conjugates for double labeling.
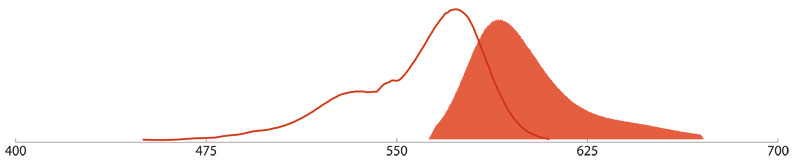
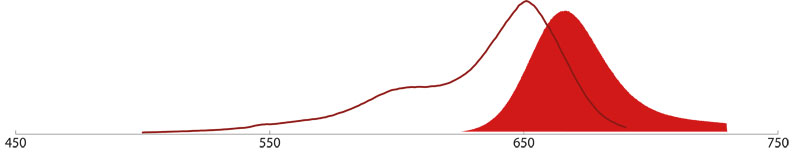
R-Phycoerythrin (R-PE), Allophycocyanin (APC), and Peridinin-Chlorophyll (PerCP)
R-PE (240 kDa), APC (104 kDa), and PerCP (35 kDa) belong to the light-harvesting phycobiliproteins, a family of fluorescent macromolecules in the photosynthesis systems of different algae. As phycobiliproteins contain more than 30 covalently linked tetrapyrrole pigments, the so-called phytochromobilins, their fluorescence signals are very intense. In the photosynthesis systems of the algae, these molecules serve to absorb and transmit light (energy) by fluorescence resonance energy transfer (FRET) to chlorophyll molecules located in the photosynthetic reaction centers. The arrangement of the pigments in the protein is naturally optimized for maximal light collection and fluorescence combined with minimum quenching caused by internal or external influences.
For this reason, after phycobiliproteins are conjugated to highly adsorbed secondary antibodies, there is little fluorescence quenching (reduction of signal strength) compared with conventional fluorophore-antibody conjugates.
Antibody conjugates of phycobiliproteins show a very high specific fluorescence activity at high extinction coefficients and thus very high quantum yields. The fluorescence of some phycobiliproteins is approximately equivalent to about 30 fluorescein or 100 rhodamine molecules at comparable excitation. Phycobiliprotein conjugates can be excited by light over a wide range of the visible spectrum and are characterized by large Stokes shifts of their emission maxima to longer wavelengths. Further advantages of phycobiliprotein conjugates include their excellent stability, the high water solubility and the insensitivity to changes in pH value. These characteristics, in combination with the high fluorescence intensity, have been shown to be most useful in laser excitation-based techniques such as flow cytometry.
PerCP, Alexa Fluor 488 (or FITC), and R-PE are excited at 488 nm with an argon laser, and thus can be used for one- to three-color analyses with single-laser flow cytometers. APC and Alexa Fluor 647 are excited at 633 or 635 nm to give a fourth color with dual-laser flow cytometers.
It should be noted that the relatively high molecular weights of PerCP, R-PE, and APC may preclude their use in procedures requiring good penetration into cells and tissues. They are predominantly intended for surface labeling of cells for flow cytometry.
R-PE and APC can be excited by light over a wide range of the visible spectrum, are highly water soluble, have relatively low isoelectric points, and lack potentially sticky carbohydrates. For this reason R-PE and APC are particularly useful for applications where either a high sensitivity, an excellent discrimination, or a multicolor labeling is required.
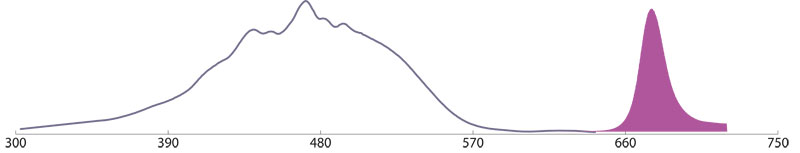
R-Phycoerythrin (R-PE)
The 240 kDa Phycobiliprotein R-PE is isolated from red macrophytic algae (seaweed). The excitation maximum of R-PE is at 490 nm a lower adsorption peak at 545/566 nm and emits maximal fluorescence at 580 nm.
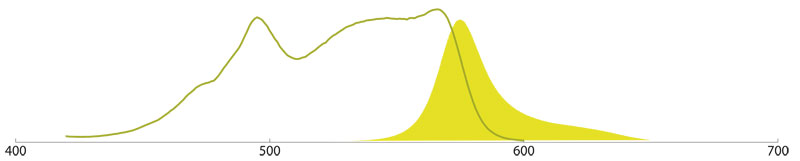
Allophycocyanin (APC)
APC is isolated from the blue-green alga Spirulina, and is chemically cross-linked for stability. The phycobiliprotein contains the chromophore Phycocyanobilin and shows a maximum excitation at 650 nm and maximum emission peak at 660 nm.
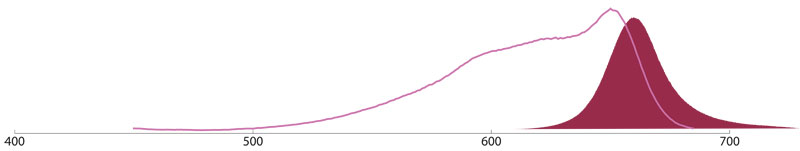
Peridinin-Chlorophyll (PerCP)
PerCP is a fluorescent peridinin-chlorophyll protein complex isolated from dinoflagellates (Dinophyceae sp.) with a molecular weight of about 35.5 kDa. It has a broad spectrum of excitation with a main peak at 472 nm, and a long Stokes shift to an emission peak at 677 nm.
Alexa Fluor®, Rhodamin Red™-X and Texas Red® are registered trademarks of Life Technologies, Inc.*. Cy™ is a trademark of GE Healthcare (Patent 5.268.486). Brilliant Violet™ is a trademark of Sirigen Inc., a Becton, Dickinson and Company affiliate. DyLight™ Fluorescent Dyes is a trademark of Thermo Fisher Scientific. The use, manufacture, trading, or sale of these dyes or products containing these dyes is sold pursuant to license agreements of Jackson ImmunoResearch Laboratories, Inc. with the mentioned companies for use of their fluorescent dye technologies.
Image courtesy of Jackson ImmunoResearch
